Recently, the international sleep respiratory field has witnessed an important technological breakthrough—a new generation of PAP technology centered on dynamic pressure regulation has received approval from US regulatory authorities. This marks a new stage in technological exploration of respiratory therapy devices in terms of comfort and long-term compliance.This progress not only reflects the global development trend of respiratory therapy technology, but also provides clearer technical direction for enterprises that have long focused on R&D and manufacturing in this field.
As an enterprise that has long focused on R&D of respiratory therapy devices, Beyond Medical has always carried out technical layout around “long-term user usage scenarios”. Under its R&D system, Beyond Medical continues to invest in real-time sensing and analysis of multi-dimensional respiratory parameters, refined and phased control of pressure output strategies, and personalized adaptation for different user groups and treatment stages. Along this technological evolution path, Beyond Medical has participated in and deeply developed a new generation of KPAP technical solution based on the concept of dynamic pressure regulation.

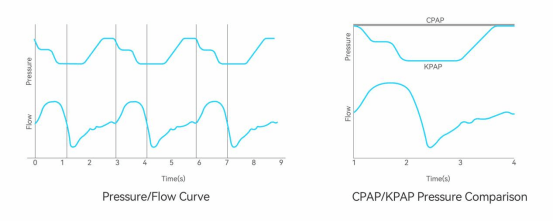
KPAP stands for Kairos Positive Airway Pressure, a dynamic pressure regulation technology based on respiratory cycles, with the following main features:
Dynamic Pressure Regulation
1.Unlike traditional CPAP which provides constant positive pressure, KPAP dynamically adjusts pressure based on changes in respiratory cycles
2.Provides necessary support only during critical respiratory phases when the airway is most prone to collapse, avoiding unnecessary pressure burden
Physiological Rhythm Alignment
1.Pressure output timing is synchronized with the body’s natural respiratory rhythm, making inhalation and exhalation more comfortable
2.Reduces nighttime respiratory discomfort and mask resistance through a more natural airflow experience
Improved Long-Term Usage Experience
1.Refined pressure control helps improve mask seal, reducing air leakage and related discomfort
2.Enhances nighttime usage comfort, thereby improving long-term treatment compliance
Efficacy and Safety Assurance
1.Achieves treatment efficacy comparable to traditional PAP therapy while ensuring stable airway patency
2.Provides a physiological-feature-aligned technical solution for long-term non-invasive respiratory therapy
Facing International Standards, Steadily Advancing Globalization Process
Against the backdrop of increasingly stringent global regulatory systems, new generation respiratory therapy technologies impose higher requirements on safety, stability, and system reliability.
Beyond Medical’s related products have passed audits by international regulatory systems including the US FDA, and are continuously advancing product iteration and technological upgrading in accordance with international market standards, gradually improving global product and quality system construction.
In the future, as products are further implemented in overseas markets, Beyond will also integrate usage feedback accumulated from different markets and populations back into its R&D system, forming more mature and universally applicable respiratory therapy solutions.



